Quick Summary:
- Lowers Medicare beneficiaries’ out-of-pocket costs for a broad set of plan-formulary insulins to a maximum $35 co-pay per thirty-day supply
- A voluntary model that plan sponsors may offer in their Part D enhanced plans
- CMS offering additional risk corridor protection to plan sponsors
Deeper Dive:
CMS announced the Part D Senior Savings Model, a voluntary model that enables participating Part D enhanced plans to lower Medicare beneficiaries’ out-of-pocket costs for a broad set of plan-formulary insulins to a maximum $35 co-pay per thirty-day supply throughout the benefit year.
Insights:
Medicare Part D enrollees are notoriously premium-sensitive. In 2020, average monthly premiums in Part D are $32.09 for a basic plan and $49.32 for an enhanced plan. Today, about 80 percent of PDPs are enhanced plans, with 54 percent of beneficiaries enrolled in these plans (Medicare Advantage (MA) can be considered an enhanced plan). Stemming from the premium sensitivity in the Part D/MA market, a plan sponsor was never going to shoulder the risk of increasing premiums (even slightly) to offer this type of capped insulin program. Hence, regulation was the only facilitative apparatus to accommodate meaningful insulin cost-sharing support in the coverage gap phase.
In the current Part D design, the manufacturer provides a 70% discount on branded drugs during the coverage gap phase (plan pays 5%; patient pays 25%). If a plan were to provide cost-share assistance to the patient during this phase, that assistance would be considered a supplemental benefit. Currently, the manufacturers’ 70% coverage gap discount is not provided until after the plans’ supplemental benefits are applied, thus discouraging plans from providing any support.
The new rule would allow the manufacturer discount to be applied before plan sponsor assistance, subsequently allowing plans to reduce patient cost-share without losing the enormous value of the manufacturer discount.
With manufacturers already providing the coverage gap discount, the impact of this rule is expected to be a relatively modest increase in adherence to insulin therapy from the reduced cost-sharing. Eli Lilly, Novo Nordisk, and Sanofi are all reported to be participating in the program.
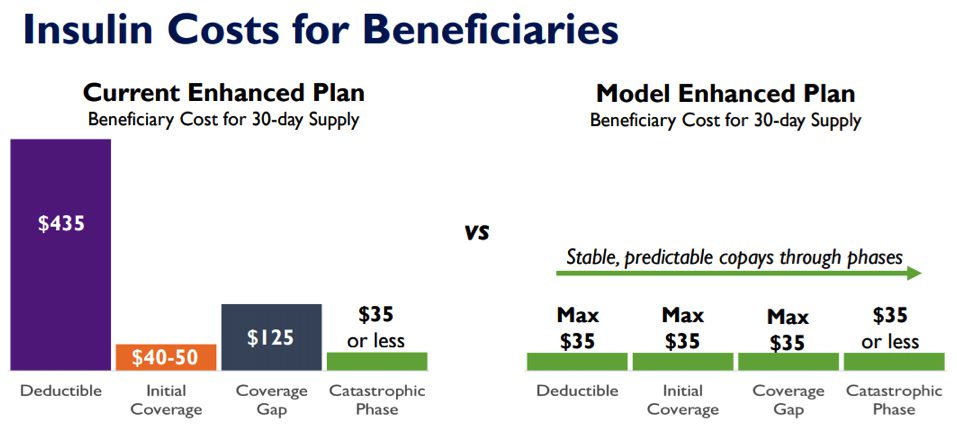
Source: CY 2021 Health Plan Innovation Models
Implementation:
CMS estimates a ~$1 PMPM increase in premiums for enhanced plans offering the program. The challenge will remain with the sponsor in effectively advertising this program within their plan offerings. Additionally, to encourage broad Part D sponsor participation, CMS is providing the option of additional risk corridor protection for 2021 and 2022 in those plans that may incur higher enrollment of insulin-dependent patients.
Timing:
The Senior Savings Model will be an option for plan sponsors and beneficiaries in the upcoming 2021 plan year.
The success, or lack thereof, with this program, could influence CMS’ potential regulation for additional highly rebated products in the part D space (i.e. asthma/COPD, DOAC’s). Naturally, this regulation comes as CMS has renewed their proposed axing of the rebate safe harbor agreement as discussed in our blog on the recent Presidential Executive Order 13939. If that proposal is not implemented, CMS may consider additional point solutions in Medicare, such as this program, to address the impact highly rebated medications have on seniors’ out-of-pocket expenses.
Viking Healthcare Solutions has established itself as the premier provider of corporate account services and strategic planning support for the pharmaceutical and biotech industries. VHS Insights, our research division, specializing in payer profiling, market research and analytics, can help you find answers to inform your payer strategy. We support traditional and rare/specialty organizations to create, maintain, defend, and protect access to your product throughout its lifecycle. Bank on our experience to help you achieve successful product commercialization. Contact us at http://www.vikinghcs.com/connect.

